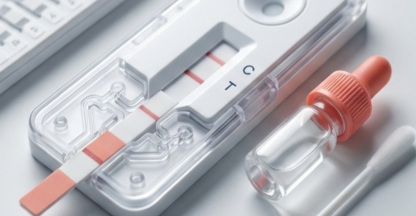
In-Vitro
Diagnostics (IVD)
Early detection enables better clinical decisions.
Comprehensive IVD solutions designed for rapid, accurate, and reliable clinical diagnostics.

Early detection enables better clinical decisions.
Comprehensive IVD solutions designed for rapid, accurate, and reliable clinical diagnostics.
From sample collection to clinical decision — our IVD systems are designed for speed, accuracy, and ease of use at every step.
Blood, urine, or swab samples are collected using sterile collection devices for analysis.
The sample is applied to the diagnostic device, initiating the detection reaction within the matrix.
Biomarkers are detected through lateral flow, electrochemical, or molecular amplification methods.
Clear, readable results enable immediate clinical decisions — at the point of care or in the laboratory.
For the qualitative and quantitative detection of specific antigens, antibodies, metabolites, and nucleic acids in human biological samples — supporting diagnosis, monitoring, and management of infectious, metabolic, and chronic diseases
“Discover our advanced IVD technologies designed for rapid, accurate, and reliable clinical diagnostics.”